Fabiosys Innovations is redefining the control of infections in the Indian healthcare realm. Conceived at IIT Delhi and fuelled by the collective brilliance of engineers, doctors, and visionaries from IIT Delhi and AIIMS, the startup pursues a clear mission: to make world-class healthcare not aspirational, but accessible.
Its patented antimicrobial surgical supplies are not mere enhancements. They are disruptive breakthroughs: engineered to eliminate 99.9% of pathogens and rigorously validated for safety and performance. In hospitals, they translate into fewer infections, faster recoveries, and superior patient outcomes. At a national scale, they accelerate India’s march toward becoming a global healthcare hub.
Fabiosys thrives on collaboration. Supported by leading institutions, such as Pfizer, Max Hospital, AIIMS, IIT Delhi, and the Red Cross Society, it fuses medical science with engineering ingenuity. The outcome is not just problem-solving products but a bold reimagination of healthcare itself: how it looks, how it feels, and how it functions in the 21st century.
At FITT Forward 2025, hosted by FITT and IIT Delhi, Yatee Gupta, Founder and CEO of Fabiosys Innovations, shared his vision in an exclusive conversation with The Interview World. He detailed the science behind Fabiosys’ antimicrobial textiles for cutting down infections and their proven efficacy in clinical settings. He also offered rare insights into the commercialization journey, spotlighted the pipeline of new innovations, and laid out his roadmap for global expansion.
Here are the key takeaways from his powerful dialogue.
Q: Could you elaborate on the antimicrobial textiles developed by Fabiosys and explain their proven efficacy in reducing infections in real-world clinical settings?
A: We develop anti-infective surgical supplies that directly tackle one of the most pressing challenges in Indian healthcare: surgical site infections. Step inside a typical operation theatre, and the problem becomes clear. Many such theatres across India remain heavily contaminated.
The consequences are staggering. According to an ICMR study, as many as 40% of patients undergoing surgery in these settings contract infections from their surroundings. That figure is not just alarming. It is catastrophic. To confront this crisis, we have created a new generation of infection-resistant surgical supplies.
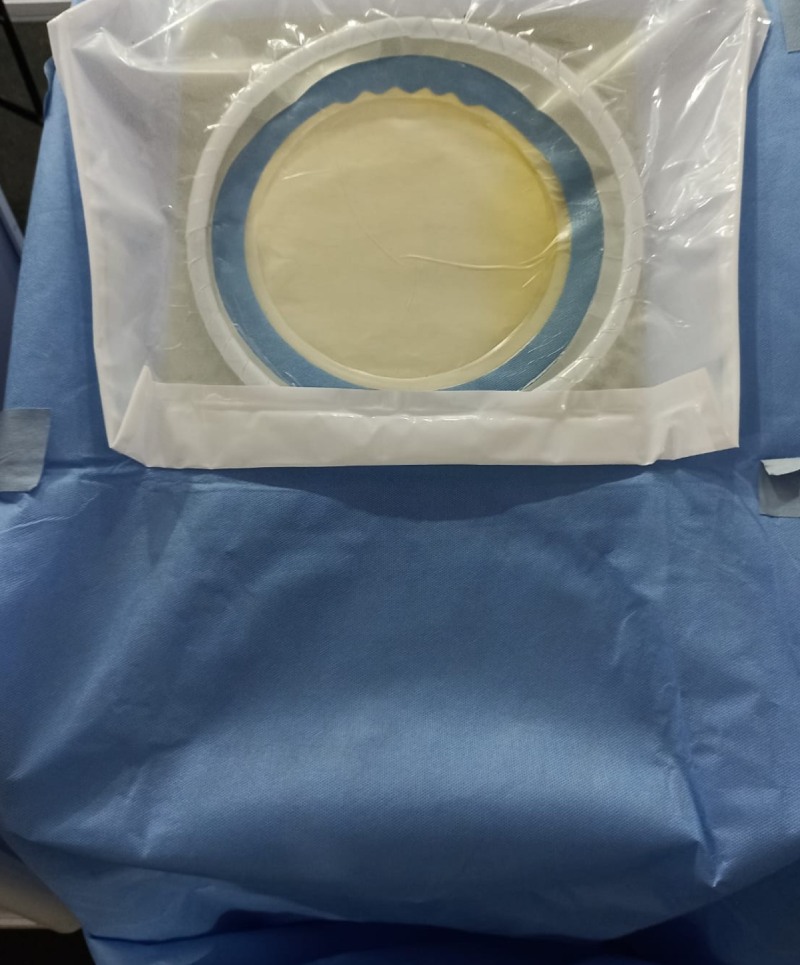
Our flagship product is the surgical drape. Picture an operating theatre: the patient lies on the table, covered by a wide sheet of blue fabric. That fabric is the surgical drape. It shields the patient and the incision area during surgery. But here lies the problem: throughout an operation, numerous healthcare professionals enter and exit the theatre. They touch equipment, surfaces, and instruments. With every interaction, the risk of cross-contamination multiplies. In long procedures, sometimes lasting 24 to 36 hours, traditional infection control practices simply cannot preserve sterility.
This is where our innovation changes the equation. We design anti-infective textiles that actively neutralize pathogens. Their unique property ensures that over 99.9% of bacteria, viruses, and fungi landing on the fabric are destroyed instantly. As a result, the surgical field remains decontaminated for the entire duration of the procedure, whether it lasts a few hours or more than a day.
The benefits extend far beyond the operating table. Patients recover faster and with fewer complications. Under current protocols, patients must return after 7 to 30 days for post-surgery inspections. When infections occur, many require readmission. The situation is especially severe when implants are involved. If an implant site becomes infected, the implant often needs to be removed, forcing additional surgeries with devastating consequences.
Our anti-infective surgical drapes prevent such outcomes. By maintaining sterility and eliminating pathogens at the source, they not only protect patients during surgery but also dramatically reduce complications afterward. In short, they transform infection control and redefine surgical safety.
Q: Have these antimicrobial textiles been commercialized yet, and if so, could you share which hospitals or healthcare institutions are currently using them?
A: I cannot disclose the names just yet, as the orders are still being finalized. However, we are already conducting paid pilot trials in some of India’s leading hospitals. These include both private and public institutions.
Our samples have been dispatched, tested, and approved. In fact, they have already been used on patients—and the outcomes so far are highly encouraging.
That said, we remain in the trial phase. Once the pilots transition into full-scale orders, we will be in a position to reveal the names and share the impact more openly.
Q: What new innovations or advancements do you envision building over the next 5 to 10 years to expand upon your existing products?
A: Our innovation falls under the category of medical textiles. At first glance, it looks like an ordinary fabric. Yet, from a regulatory standpoint, it qualifies as a Class A medical device. That classification changes everything.
Unlike conventional textiles, a Class A medical device must pass stringent checks and validations. Every product must demonstrate absolute safety before reaching patients.
We subjected our fabric to rigorous testing. For instance, we conducted skin sensitivity tests to confirm that it does not trigger allergic reactions or harm human skin. We also carried out animal trials on white rabbits, which verified the absence of skin irritation. These protocols ensured that our material meets the highest safety benchmarks.
The results speak for themselves. Our fabric has been proven biocompatible, with certifications from SITRA and validations from other international laboratories. In short, it is more than a textile. It is a medical-grade solution built on scientific rigor and regulatory compliance.
Q: Do you plan to take these products to international markets, or will your focus remain primarily on the domestic market?
A: We have already dispatched our samples to more than 14 countries. These include several African nations such as Nigeria and Ethiopia, as well as markets like Singapore, the UAE, and Dubai. While these engagements remain at an early stage, they mark the beginning of our international journey.
Our immediate priority is clear: establish a strong foothold in the Indian market first. Only then will we scale abroad. As a startup, we believe in building step by step, with a focus on sustainable growth.
To assess global demand, we shipped samples through our distributor network. The feedback, particularly from Africa, has been overwhelmingly positive. The region faces unique healthcare challenges. Unlike COVID-19, which dominated global headlines for two years, many African countries battle localized outbreaks almost every six months, yet these rarely attract international media attention.
This is where our solution becomes critical. We are actively engaging with leading non-profits, including Doctors Without Borders (MSF). Our discussions center on piloting our products in hospitals across low-resource settings, where affordability is a barrier for patients but where governments, NGOs, and even the UN can intervene.
In parallel, we continue to push forward in India with full intensity. For us, the mission is twofold: strengthen infection control at home and extend meaningful impact to vulnerable healthcare systems abroad.