Haomagraft, Gujarat’s first licensed bone bank, transforms donated bone into life-changing allografts. Every graft undergoes rigorous donor screening, validated sterilization, and precise preservation because safety is not optional, it is sacred. From femoral heads to demineralized granules, each product empowers orthopedic surgeons to rebuild bones, restore function, and save lives. Guided by expert leadership and a vision to expand to 100 cities nationwide, Haomagraft seamlessly integrates science, ethics, and impact. This is more than tissue banking; it is a promise: unwavering standards, clinical reliability, and a commitment to every patient whose life depends on each graft delivered.
In an exclusive conversation with The Interview World at Expodent International India 2025, Sanket Shah, Director, and Hufreez Kasad, Director of Haomagraft, shared how the company preserves bone and develops diverse products. They offered insights into market response, explained the principles behind their products, highlighted adherence to international standards, and emphasized essential quality parameters.
Here are the key takeaways from their compelling discussion.
Q: How is Haomagraft advancing efforts to preserve and regenerate bone?
A: We operate a bone bank based in Ahmedabad, Gujarat. Our primary focus is procuring bones from orthopedic operating theaters, particularly during knee and hip replacement surgeries. Bones that would otherwise be discarded as biohazard waste are carefully collected and brought to our licensed processing facility, which operates under the Transplantation of Human Organ Act and the state authority for organ transplantation.
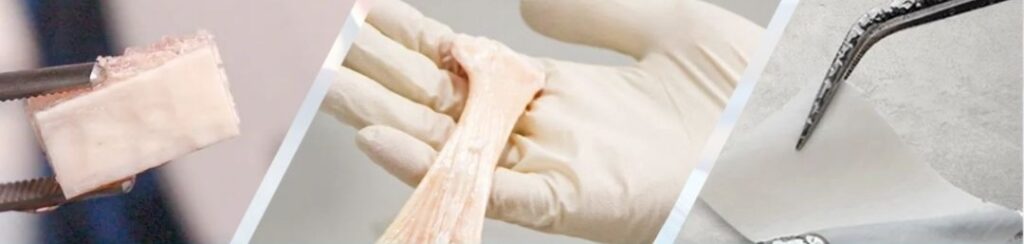
At our facility, we process these bones to create safe, reusable allografts. Unlike synthetic or ceramic implants, which the body often perceives as foreign, our products integrate naturally and are fully accepted by the body. As a result, patients experience complete recovery after implantation, with outcomes that are reliable and long-lasting.
Over the past three years, we have supported more than 3,000 surgeries using over 5,000 grafts, achieving a 100% success rate, no rejections, no infections, and no adverse effects. Our allografts are now widely used across orthopedic, dental, oncology, neurology, spine, and trauma surgeries. In these six medical segments, doctors consistently rely on our products to deliver predictable, high-quality patient outcomes.
Q: How many products have you developed to date?
A: Currently, we offer three core products across different categories. The first, focused on dental applications, includes demineralized bone granules such as Demineralized Freeze-Dried Bone Allograft (DFDBA) and Demineralized Bone Matrix (DBM). The second category consists of allograft chips, and the third is femoral heads. All three products are processed to achieve complete sterility, adhering strictly to global standards set by the American Association of Tissue Banks.
Once prepared, these products are delivered to surgeons and doctors, who use them in a variety of procedures. Patients are experiencing outstanding outcomes, with results that consistently demonstrate the efficacy and safety of our allografts.
Q: What kind of response have you received to these initiatives across India?
A: We are receiving strong support from doctors, manufacturers, and distributors across India. While the demand for these products has always been recognized, the supply was previously limited, requiring imports. By developing these allografts domestically, we have not only advanced the Make in India initiative but also created a reliable import substitute.
Today, our products are widely used, with a presence in over 18 states and more than 70 cities. Leading corporate hospitals and prominent dental chains across the country rely on our allografts regularly, reflecting the trust and acceptance we have earned among medical professionals.
Q: Could you elaborate on the principles it operates on?
A: Our guiding principle is simple: everything that nature gives us can serve a purpose. While recycling is common for many natural resources, the potential to recycle human organs has remained largely untapped until now. We focus on recovering biological material that would otherwise be discarded, reducing potential environmental impact.
By procuring and processing this material, we transform what was once considered waste into life-changing allografts. The result is a remarkable product that directly benefits patients, turning what could be lost into a source of healing and hope.
Q: Since this concept is new in the country, what practices exist internationally regarding it?
A: Several developed countries already maintain well-established bone banks. While the concept is not entirely new, it holds significant potential for growth. The United States, Japan, the United Kingdom, Singapore, and Australia, among others, have successfully implemented comprehensive bone banking systems, demonstrating both the feasibility and the value of this practice.
Q: Are there any specific parameters that a recipient must meet?
A: For the recipient, our allografts act as a biocompatible material within the body. During production, we rigorously monitor quality control parameters, strictly following the guidelines of the American Association of Tissue Banks (AATB).
When we receive a bone from a donor, we also collect a blood sample and conduct additional mandatory tests, beyond the preoperative tests performed by the physician. These include HIV, Hepatitis B and C, and syphilis, ensuring safety at the source.
Once the graft is processed, we perform further testing on the finished product, including assessments of bioburden and moisture content. This multi-layered approach guarantees that every graft meets the highest standards of safety and efficacy before it reaches a patient.









1 Comment
I wish I had read this sooner!
Comments are closed.